
Our News
Alnylam Announces Major Expansion of U.S. Pharmaceutical Manufacturing Facility
$250 million investment will add new capacity and siRNA enzymatic ligation capabilities to Alnylam's Norton, Massachusetts site
December 17, 2025
Alnylam has announced a major expansion of our state-of-the-art pharmaceutical manufacturing facility in Norton, Massachusetts. This 200,000 sq. ft. facility, which opened in 2021, is the center of Alnylam's global RNAi therapeutics manufacturing operations and produces both clinical and commercial supply of siRNA oligonucleotide drug substance for patients around the world.
This investment of $250 million will expand capacity at the Norton site significantly reducing production costs, and position Alnylam to support future launches across our robust and growing pipeline. The expansion will build what is poised to become the industry’s first fully dedicated, proprietary, siRNA (small interfering RNA) enzymatic-ligation manufacturing facility.
At this pivotal time with our expanding pipeline of RNAi therapeutics, Alnylam is accelerating development of siRNA manufacturing and changing what’s possible in a single facility. This advance will be a critical enabler in the scaling of our pipeline to include potential treatments for diseases such as hypertension, type 2 diabetes, and obesity.” - Yvonne Greenstreet, M.D., MBA, Chief Executive Officer
siRELIS™ siRNA Enzymatic Ligation Manufacturing Platform Announcement
As part of this announcement, Alnylam also shared that our next-generation, scalable, siRNA enzymatic ligation manufacturing platform, siRELIS™, has been accepted into the U.S. Food and Drug Administration’s (FDA) Emerging Technology Program, accelerating dialogue with global health authorities on innovative manufacturing approaches for oligonucleotide-based medicines. This acceptance follows successful demonstration of Alnylam’s enzymatic ligation platform through production of pilot-scale batches of zilebesiran and nucresiran.
Read the full press release here.
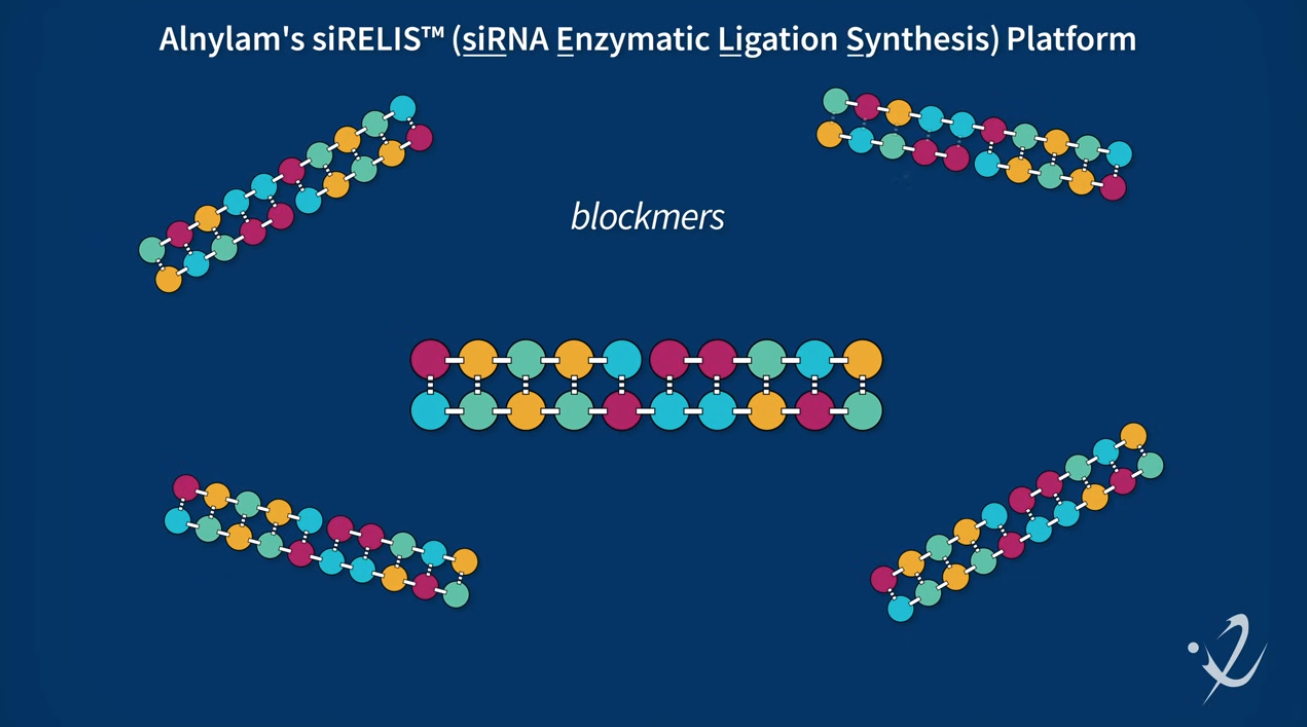 Alnylam's siRELIS siRNA enzymatic ligation manufacturing platform explained
Alnylam's siRELIS siRNA enzymatic ligation manufacturing platform explainedNorton Expansion Groundbreaking Event
To celebrate the announcement, Alnylam hosted a groundbreaking event with over 150 employees, partners and distinguished guests from government and industry. The groundbreaking featured remarks from our CEO Dr. Yvonne Greenstreet, Massachusetts Governor Maura Healy, Massachusetts Live Sciences Center (MLSC) President and CEO Dr. Kirk Taylor, and Massachusetts Secretary of Labor and Workforce Development Lauren Jones.
 Alnylam's Norton groundbreaking event recap
Alnylam's Norton groundbreaking event recapWe invite you to learn more about Alnylam and our pioneering work in RNAi therapeutics, the Science of RNAi and how we make our medicines.
Tags
RNAi, Articles, Manufacturing, Norton, Our Science, RNAi, siRNA, siRELIS, Enzymatic Ligation





